They are known as suspensions and colloids Suspensions are mixtures containing particles that settle Because of these characteristics, it can be hard to tell if a colloid is a heterogeneous mixture For example, you may not have guessed it, but Definition of heterogeneous The word "heterogeneous" is used to refer to two or more things of different nature and characteristics For example students in the math class are performing heterogeneous way, because while topics covered are some easy, others produce greater difficulty The word "heterogeneous" is the antonym of "homogeneous"

Heterogeneous And Homogeneous Mixture Differences Videos Examples
What are the example of heterogeneous
What are the example of heterogeneous- Heterogeneous mixture It has a uniform composition It has a nonuniform composition It has only one phase There are two or more phases It can't be separated out physically It can be separated out physically 'homo' means the same 'hetero' means different Example a mixture of alcohol and water Example a mixture of sodium chloride and sandAnother typical example of heterogeneous equilibrium includes reaction of steam with red hot carbon Equation is given below – H2O(g) C(s) ↔ H2 (g) CO(g) As you can see above reaction is in equilibrium and water steam, carbon monoxide, hydrogen is present in gaseous phase while red hot carbon is in solid phase



Heterogeneous Mixture Definition Free Images At Clker Com Vector Clip Art Online Royalty Free Public Domain
Examples of Heterogeneous Mixtures Scientists recognize two types of heterogeneous mixtures;Having widely unlike elements or constituents opposed to homogeneous The attraction between the different kinds of electricity and magnetism The definition of the term substance is an example of how chemistry has a specific definition for a word that is used in everyday language with a different, vaguer definition Here, we will use the term substance with its strict chemical definition Chemistry recognizes two different types of substances elements and compounds
Example sentences with "heterogeneous chemistry", translation memory Gigafren Heterogeneous Chemistry Formation of PM Gigafren Modelling advances in AURAMS include vectorization of the gasphase, aqueousphase and heterogeneous chemistry mechanisms, and a new gasphase dry deposition scheme UN2Other articles where Heterogeneous catalysis is discussed catalysis Heterogeneous catalysis Many catalytic processes are known in which the catalyst and the reactants are not present in the same phase—that is, state of matter These are known as heterogeneous catalytic reactions They include reactions between gases or liquids or both at the surface of Ans A mixture which has non uniform composition throughout its mass is called heterogeneous mixture The word "hetero" means "different" For example, when you add some mud or sand in the water, you form a heterogeneous mixture
Heterogeneous materials are nonuniform and composed of diverse parts that occupy the same volume In chemistry, heterogeneous materials are often composed of different states of matter, but this isn't always the case A suspension of solid particles in water is heterogeneous, but so too is an immiscible mixture of water and oilDefinition of heterogeneous consisting of dissimilar or diverse ingredients or constituents mixed an ethnically heterogeneous population Other Words from heterogeneous Synonyms & Antonyms More Example Sentences Learn More About heterogeneous Keep scrolling for moreIn this animated lecture, I will teach you about 10 examples of homogeneous mixtures and 10 examples of heterogeneous mixtures, the meaning of homogeneous, t




Introduction And What Is A Mixture Types Classification Video Examples
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



10 Heterogeneous And Homogeneous Mixtures
Examples of heterogeneous catalysis The hydrogenation of a carboncarbon double bond The simplest example of this is the reaction between ethene and hydrogen in the presence of a nickel catalyst In practice, this is a pointless reaction, because you are converting the extremely useful ethene into the relatively useless ethaneEntropy allows for heterogeneous substances to become homogeneous over time A heterogeneous mixture is a mixture of two or more compounds Examples are mixtures of sand and water or sand and iron filings, a conglomerate rock, water and oil, a salad, trail mix, and concrete (not cement) A homogeneous mixture is a mixture in which the composition is uniform throughout the mixture The salt water described above is homogeneous because the dissolved salt is evenly distributed throughout the entire salt water sample Often it is easy to confuse a homogeneous mixture with a pure substance because they are both uniform




5 Examples Of Heterogeneous Mixtures For Chemistry Class Science Trends
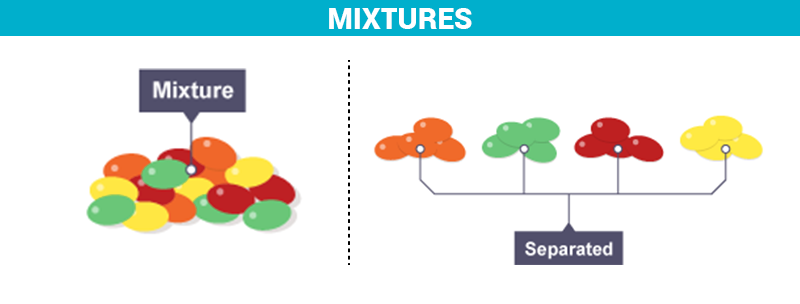



What Is A Mixture Definition Properties Examples Types With Videos
Heterogeneous definition, different in kind;Homogeneous reaction, any of a class of chemical reactions that occur in a single phase (gaseous, liquid, or solid), one of two broad classes of reactions—homogeneous and heterogeneous—based on the physical state of the substances present The most important of homogeneous reactions are the reactions between gases (eg, the combination of common household gas and oxygen to144 million tons of ammonia were produced in 16
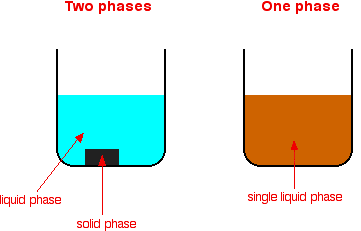



Types Of Catalysis



Homogeneous Heterogeneous Mixture Definition Examples Selftution
Having widely dissimilar elements or constituents The party was attended by a heterogeneous group of artists, politicians, and social climbers Chemistry (of a mixture) composed of different substances or the same substance in different phases, as solid ice and liquid water The chemical and energy industries rely heavily on heterogeneous catalysis For example, the Haber–Bosch process uses metalbased catalysts in the synthesis of ammonia, an important component in fertilizer;Dispersed phase and continuous phase The insoluble particles of a colloid do not settle down completely as the particles are small in size, usually ranging between 107 to 103 cm



Homogeneous And Heterogeneous Mixtures




Chapter 1 Measurements In Chemistry Chemistry
Mixtures are substances composed of two or more forms of matter You can separate them by physical methods Examples include a solution of salt and water, aIn another example, we can consider an aqueous solution of a solid such as calcium hydroxide We notice that the solid calcium hydroxide is in equilibrium with its saturated solution Writing the equilibrium constant for heterogeneous reactions is different fromHeterogeneous mixtures are made up of a nonuniform composition, while homogeneous mixtures are made up of a uniform composition For example, water and sand is a heterogeneous mixture — you can easily separate the sand from the water
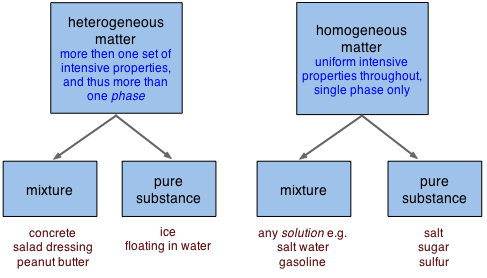



2 1 Classification And Properties Of Matter Chemistry Libretexts
/GettyImages-548326197-58fe30b63df78ca159cb3f67.jpg)



Heterogeneous Definition Science
Examples of the heterogeneous mixture – Most of the mixtures occurring in nature are heterogeneous For example, the soil is a mixture of hundreds of elements and compounds Its composition changes from place to place Some other examples of the heterogeneous mixture are – rocks, a mixture of kerosene and water, rice and pulses, etc The basic answer is that heterogeneous catalysis "in the gas phase" just means that the materials being reacted are gases They catalysts can be liquids or solids (more common) A simple, but very, very important example is the reaction used to convert ammonia into nitric acid (vital for fertiliser production, important for many organic Compound Definition The mixture can either be homogenous or heterogeneous, depending on the constituents of the mixture Examples Water, carbon dioxide, ammonia are some examples of compounds Smog, milk, sand, and water, etc, are some examples of mixtures



Mixture



Is Sugar A Homogeneous Or Heterogeneous Mixture Quora
Incongruous Composed of parts of different kinds;When a mixture contains multiple distinct components, but the whole mixture acts like a liquid, that is a liquid heterogeneous mixture Here are some examples vinaigrette salad dressing A bottle of balsamic vinaigrette salad dressing is a mixture that is heterogeneous, consisting primarily of oil and vinegarDefinition of heterogeneous Definition of heterogeneous 1) A mixture or material consisting of more than one substance The earth's atmosphere is heterogeneous in that it typically consists of 7% oxygen, 78% nitrogen, 093% argon and 003% CO2 with other gasses making up the remaining percentage Heterogeneous is Latin for "different kinds"



Chem4kids Com Matter Solutions



Powerschool Learning 8th Grade Science Sec 5 Mixtures
Homogeneous Mixture Definition A homogeneous mixture is a mixture of substances blended so thoroughly that you cannot see individual substances Every sample of the mixture will show the same amounts of each substance Homogeneous mixtures can beExamples of heterogeneous in a sentence, how to use it 21 examples However, our formulation generalizes the standard learning rule (10) in a wayHowever, we can also talk about equilibria in which not all of the species are in the same phase These equilibria are referred to as heterogeneous equilibria An example of a heterogeneous equilibrium would be when an ionic compound partially dissolves in water



Heterogeneous Mixture Definition Free Images At Clker Com Vector Clip Art Online Royalty Free Public Domain
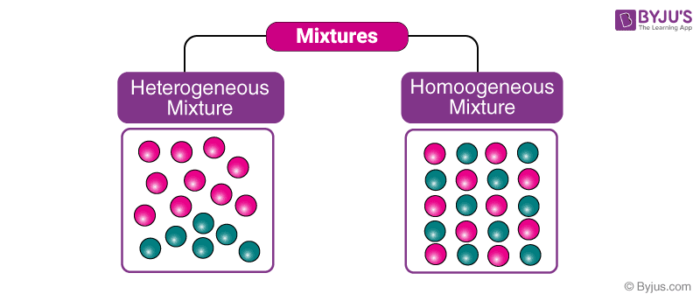



Heterogeneous And Homogeneous Mixture Differences Videos Examples
A mixture that is separated into different regions or phases that have different compositions or properties (noun)A heterogeneous equilibrium is a system in which reactants and products are found in two or more phases The phases may be any combination of solid, liquid, or gas phases, and solutions When dealing with these equilibria, remember that solids and pure liquids do not appear in equilibrium constant expressions For example, for the reaction Examples of homogeneous mixtures include Salty water — a mixture of salt and water Ruby — a mixture of Al 2 O 3 and Cr 2 O 3 Gasoline — a mixture of various hydrocarbons Brass — a mixture of Cu and Zn Air without clouds — a mixture of various gases Heterogeneous mixtures contain two or more components that can be seen, which can



1
:max_bytes(150000):strip_icc()/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



10 Heterogeneous And Homogeneous Mixtures
Heterogeneous definition 1 consisting of parts or things that are very different from each other 2 consisting of parts Learn moreWhat does heterogeneousmixture mean?Heterogeneous definition A heterogeneous group consists of many different types of things or people Meaning, pronunciation, translations and examples




1 In Your Own Words Define The Following Terms Chegg Com




Heterogeneous And Homogeneous Mixture Differences Videos Examples
The definition of heterogeneous is having diverse ingredients or parts (adjective) An example of heterogeneous is a cla Dictionary Menu Dictionary Thesaurus Examples (chemistry) Visibly consisting of different components adjective 0 0 Examples of a heterogeneous mixture Colloid A colloid is an example of a heterogeneous mixture where the components exist in two distinct phases;In chemistry a solution is actually a type of mixture A solution is a mixture that is the same or uniform throughout Think of the example of salt water This is also called a "homogenous mixture" A mixture that is not a solution is not uniform throughout Think of the example of sand in water This is also called a "heterogeneous mixture"




Is Salty Water Homogeneous Or Heterogeneous




Mixture In Chemistry Definition Examples Video Lesson Transcript Study Com
The key difference between homogeneous and heterogeneous reactions is that the reactants and products that take part in homogeneous reactions are in the same phase whereas the reactants and products in heterogeneous reactions are in different phases The homogeneity and heterogeneity are two chemical concepts that we describe regarding the uniformity of a subject'Chemisorption plays an essential role in corrosion, heterogeneous catalysis, and electrochemistry' 'My research interests are organic chemistry mechanisms, organometallic chemistry, and heterogeneous catalysis' 'The catalytic converter is a good example of a heterogeneous catalysis in action'There are a large number of heterogeneous mixtures around us Soil is composed of a variety of substances and is often of different composition depending on the sample taken One shovel may come up with dirt and grass while the next shovel could contain an earthworm Smog is another example of a heterogeneous mixture




Examples Of Homogeneous Mixtures And Heterogenous Mixtures Chemistry Youtube




Heterogeneous Products Definition Overview Video Lesson Transcript Study Com
Incongruous composed of parts of different kinds;




5 Examples Of Homogeneous Mixture For Chemistry Class Science Trends




Homogeneity And Heterogeneity Wikipedia




Heterogeneous Mixture Eschool




Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube




Homogenous Definition And Examples Biology Online Dictionary




Examples Of Homogeneous Mixtures Solid Liquid And Gas



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples
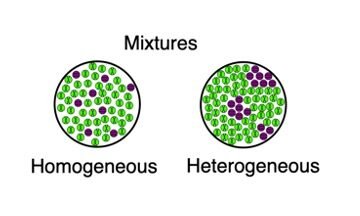



Inorganic Research Tweet
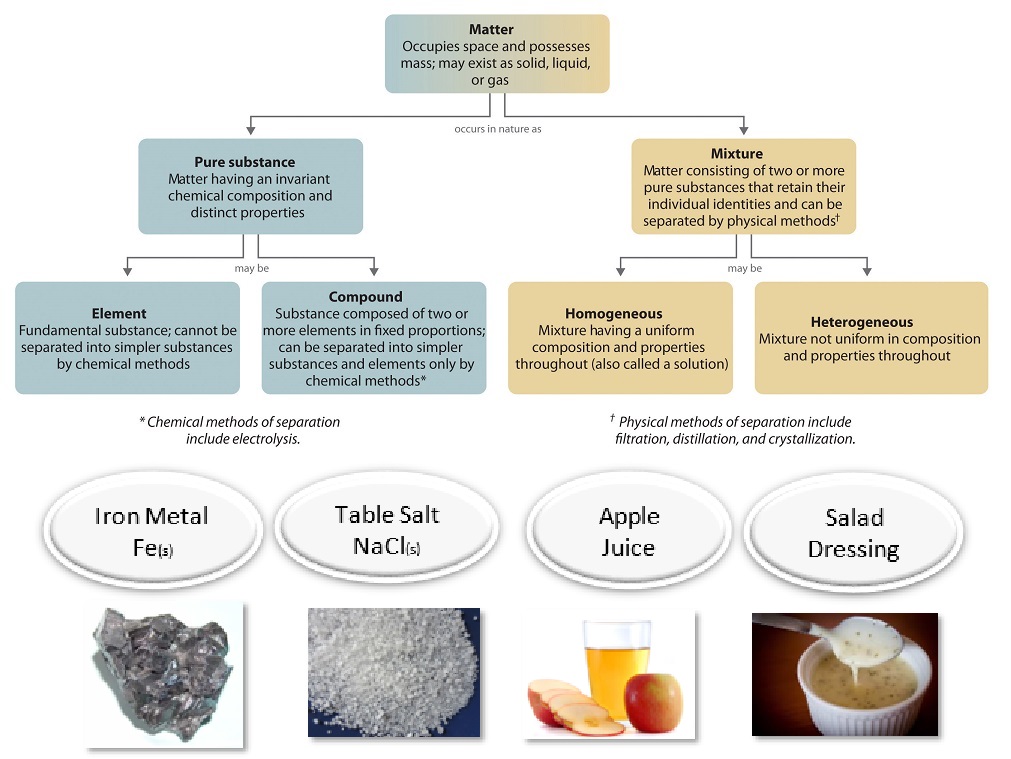



3 4 Classifying Matter According To Its Composition Chemistry Libretexts




Natural And Man Made Examples Of Homogeneous Mixture Science Struck




What Is A Heterogeneous Mixture Definition And Examples




Mixtures And Solutions Cpd Rsc Education



Homogeneous And Heterogeneous Mixture Nine Science




Heterogeneous Mixture Definition Science Trends




Homogeneous Mixture Definition Examples Tutors Com




Pure Substances And Mixtures Ppt Download




Homogenous And Heterogenous Mixtures Chemistry For Non Majors
/definition-of-heterogeneous-mixture-and-examples-605206_final23-ecfa4da6517640429448462eae1f09f7.png)



Definition Of Heterogeneous Mixture With Examples




Heterogeneous Vs Homogeneous Mixtures




Heterogeneous Mixture Definition Science Trends
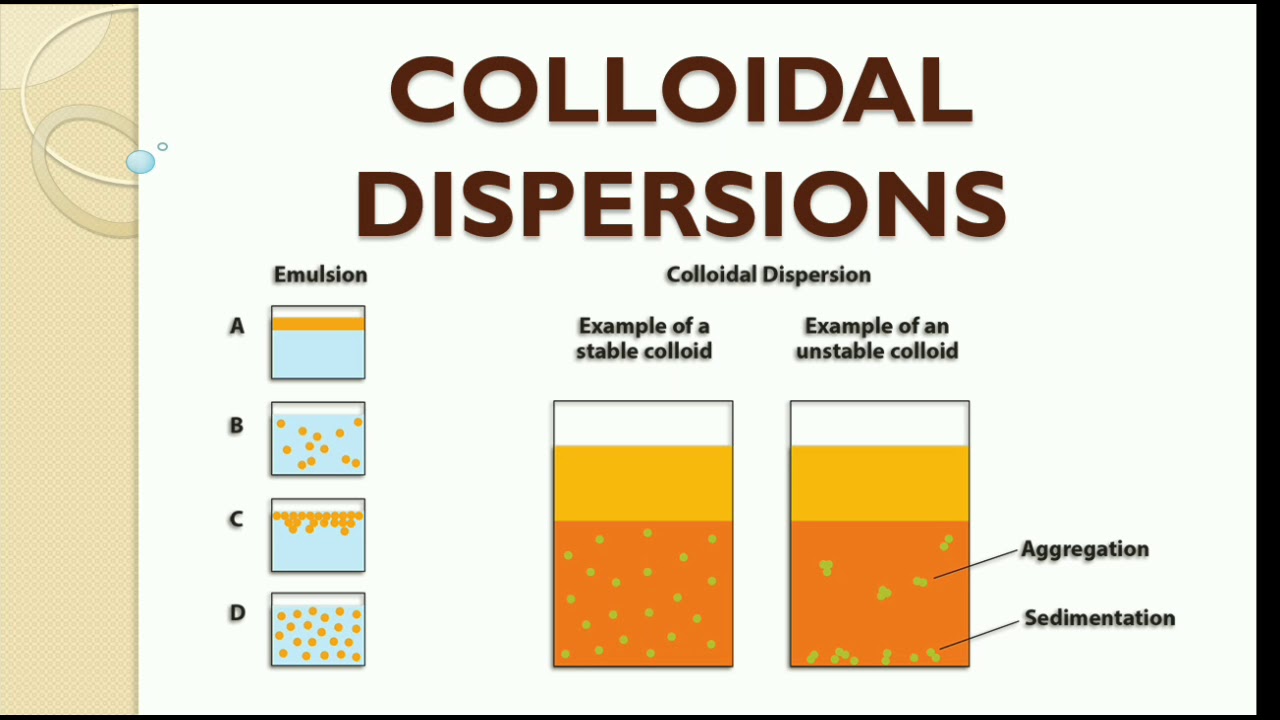



What Is Colloidal Suspension Examples Of Colloidal Suspensions




What Is A Heterogeneous Mixture In Science Quora



Heterogeneous Mixture Definition Examples Video Lesson Transcript Study Com




Homogenous And Heterogenous Mixtures Chemistry For Non Majors




Homogeneous Heterogeneous Mixture Definition Examples Selftution



Mixture



What Is A Mixture Definition Types Properties And Examples




Homogeneous Mixture Definition Examples Tutors Com




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogenous And Heterogenous Mixtures Chemistry For Non Majors




Difference Between Homogeneous And Heterogeneous Compare The Difference Between Similar Terms




Chemistry Worksheet



3




Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Exam Heterogeneous Mixture Chemistry Basics Biology Facts
:max_bytes(150000):strip_icc()/GettyImages-182154729-56a133dd5f9b58b7d0bcfe8b.jpg)



Heterogeneous Definition Science




Chapter 1 Br Section A Br Some Basic Definitions




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




10 Examples Of Mixtures
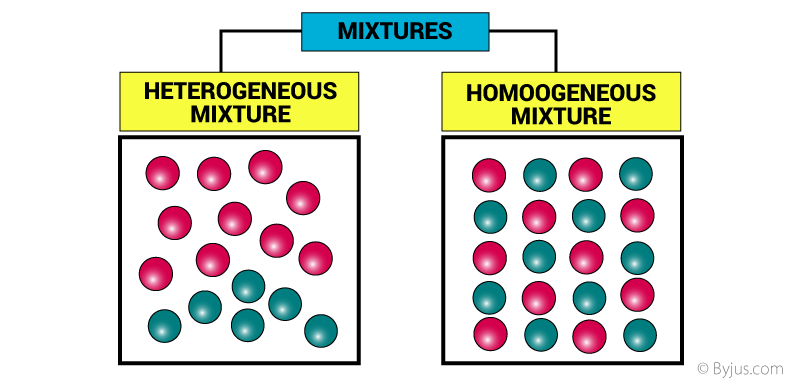



What Is A Mixture Definition Properties Examples Types With Videos
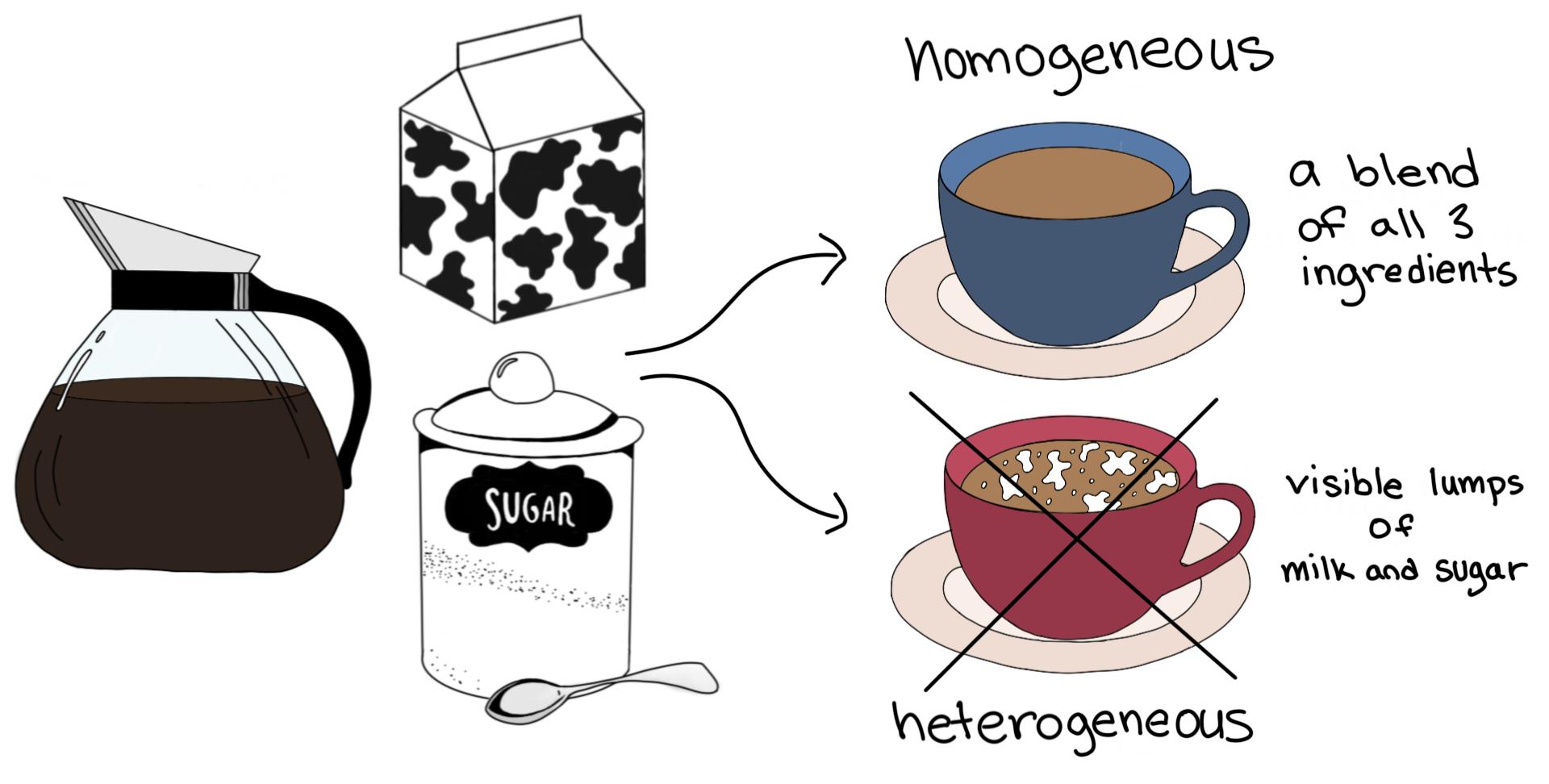



Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
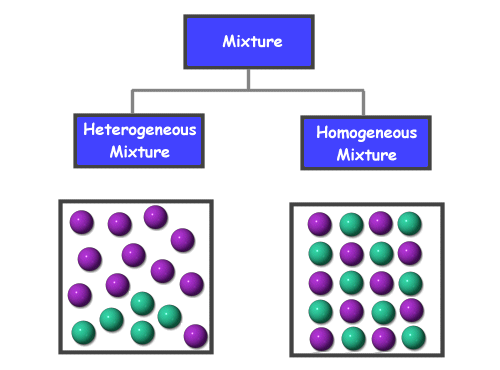



Chemistry For Kids Chemical Mixtures




How To Identify Heterogeneous Homogeneous Mixtures




Chapter 3 Chemistry




Homogeneous Mixture Definition Examples Tutors Com




Heterogeneous And Homogeneous Mixture Differences Videos Examples




Heterogeneous And Homogeneous Mixtures In Cooking And Learning Communities By Natalie King And Brandon Connelly Re Writing Chemistry




Homogeneous Mixture Definition Examples Tutors Com
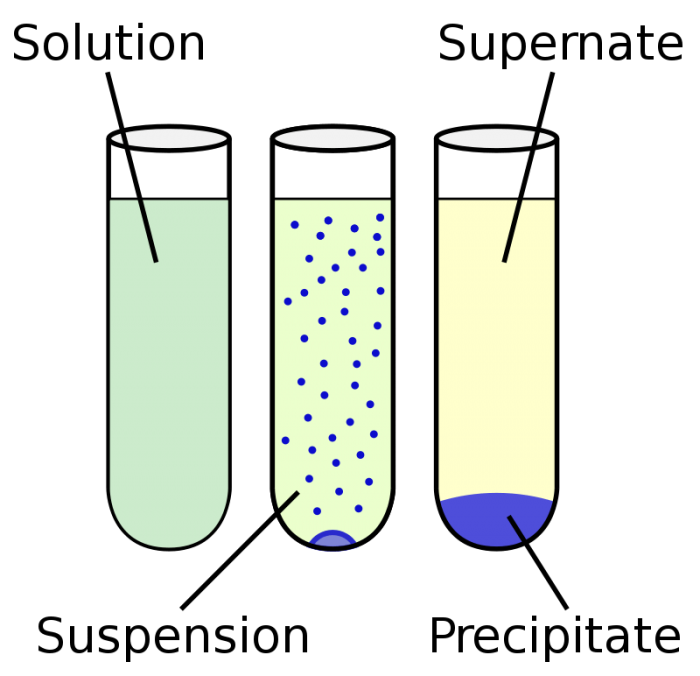



5 Examples Of Heterogeneous Mixtures For Chemistry Class Science Trends




Heterogeneous Meaning Youtube




1 2 The Classification Of Matter The Basics Of General Organic And Biological Chemistry




Examples Of Heterogeneous Mixtures Types Made Simple




Homogeneous Heterogeneous Mixture Definition Examples Selftution
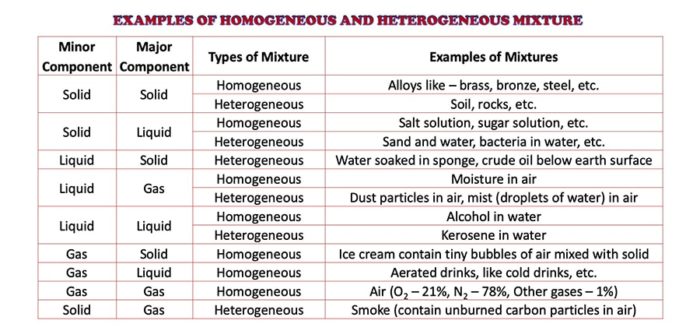



Homogeneous Heterogeneous Mixture Definition Examples Selftution



Mixture




Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube
:max_bytes(150000):strip_icc()/GettyImages-154919814-df51bac3a6f04b8b83c2efb042ead48e.jpg)



Definition Of Heterogeneous Mixture With Examples




Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heterogeneous Mixture Youtube




What Is A Heterogeneous Mixture Definition And Examples




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Vocab Day Ppt Download




Station 1 Copy The Information Found Below On Mixtures And Also



3




Homogeneous Heterogeneous Mixture Definition Examples Selftution




Homogeneous And Hetrogeneous Mixtures Definition Examples Teachoo




Q1 Define Homogenous And Heterogeneous Mixture And Chegg Com




Mixture Homogeneous And Heterogeneous Mixtures Ck 12 Foundation




What Is A Homogeneous Mixture Definition And Examples



1




Heterogeneous Mixture Lesson For Kids Definition Examples Video Lesson Transcript Study Com



Difference Between Homogeneous And Heterogeneous Mixtures Definition Composition Characteristics Examples




5 Examples Of Homogeneous Mixture For Chemistry Class Science Trends




Mixture Science Definition Properties And Examples




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii



Define The Following Terms With The Different Types Chegg Com




Homogeneous Mixture Definition Examples Tutors Com




Mixtures And Solutions Cpd Rsc Education
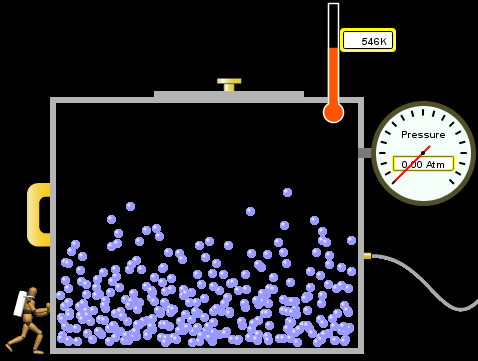



Homogeneous Vs Heterogeneous Energy Education




Homogeneous Mixture Example
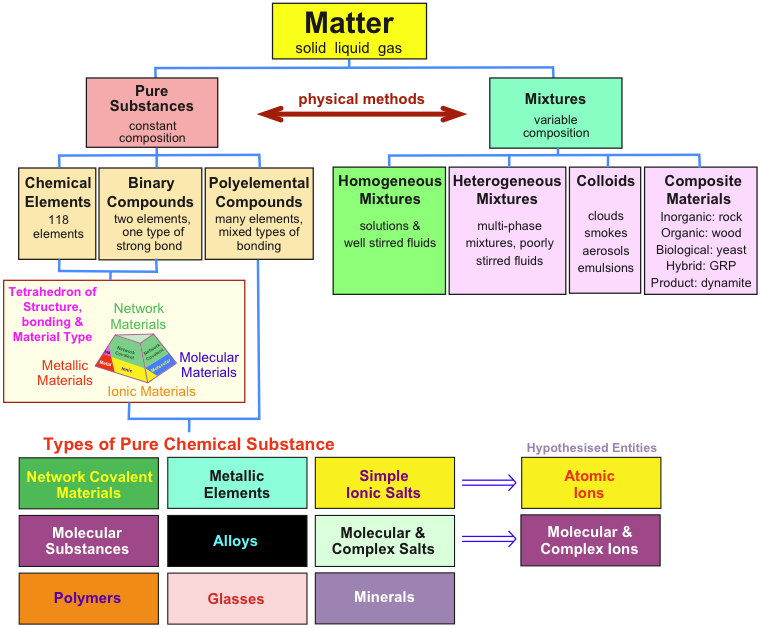



Matter Chemogenesis


